Biomedical engineering has traditionally approached implant design through the lens of biocompatibility. Materials are selected to avoid toxicity, minimize immune response, and ensure long-term chemical stability within the body. While these factors remain essential, they do not fully determine implant performance. Increasingly, clinical outcomes are shaped by a different constraint, mechanical compatibility with the surrounding biological environment.
The human body is not a static structure. Tissues deform, adapt, and respond continuously to mechanical forces. When an implant is introduced, it becomes part of this dynamic system. If its mechanical behavior does not align with the surrounding tissue, complications can arise even when the material itself is biologically inert. This has led to a growing focus on mechanical compliance matching as a central design principle in implantable devices.
The Problem of Mechanical Mismatch
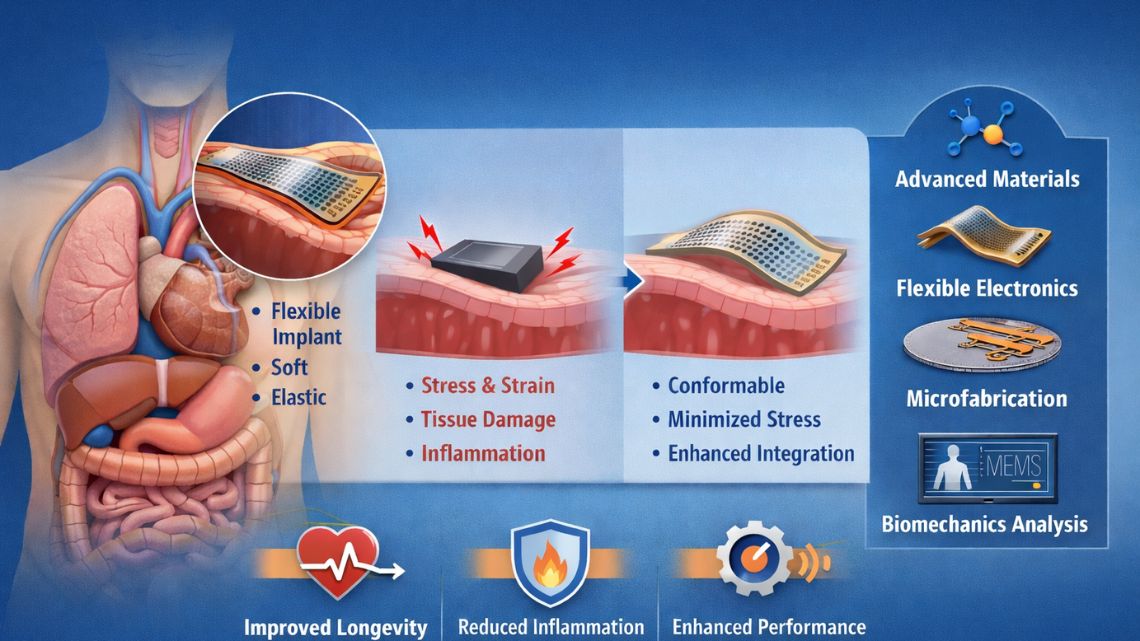
Mechanical compliance refers to how a material deforms under load. Biological tissues such as bone, muscle, and soft connective structures have specific mechanical properties that vary across locations and functions. Implants, by contrast, are often made from metals or rigid polymers with significantly different stiffness characteristics. When these materials are introduced into the body, they can alter how forces are distributed.
In orthopedic implants, for example, a rigid implant can absorb a disproportionate amount of load, reducing stress on the surrounding bone. Over time, this can lead to bone resorption, a phenomenon commonly referred to as stress shielding. In soft tissue applications, stiffness mismatch can cause irritation, inflammation, or mechanical damage. The issue is not failure in the traditional engineering sense. The implant may remain structurally intact, yet the system, comprising both implant and tissue, performs poorly.
Dynamic Loading and Long-Term Behavior
The human body subjects implants to continuous and variable loading conditions. Walking, breathing, blood flow, and even subtle movements generate forces that change over time. Unlike static engineering systems, biological environments introduce cyclic loading, micro-movements, and adaptive responses. Implants must therefore maintain performance under conditions that evolve throughout their service life.
Mechanical mismatch can become more pronounced over time. Small differences in stiffness or deformation behavior may not be immediately apparent but can accumulate into larger issues. Loosening, wear, and tissue damage often result from these long-term interactions rather than initial design flaws. This shifts the focus of biomedical engineering from immediate functionality to sustained compatibility.
Material Innovation and Structural Design
Addressing mechanical mismatch requires both material and structural solutions. Advances in biomaterials are enabling the development of implants with tailored mechanical properties. Composite materials, porous structures, and flexible polymers are being used to approximate the behavior of biological tissues.
Porous implants, for instance, reduce overall stiffness while promoting tissue integration. Additive manufacturing techniques allow engineers to control internal geometry, creating structures that balance strength with compliance.
Design strategies also play a critical role. Instead of relying solely on material properties, engineers are modifying implant geometry to influence how loads are transferred. Gradual transitions in stiffness between implant and tissue can reduce stress concentrations and improve long-term stability. These approaches reflect a shift from designing isolated components to engineering integrated systems.
Interface Behavior and Tissue Interaction
The interface between an implant and biological tissue is where mechanical compatibility is most critical. This boundary must transfer loads effectively while accommodating differences in movement and deformation. Poor interface design can lead to micromotion, which disrupts tissue integration and increases the risk of implant failure. In joint replacements, excessive movement at the interface can accelerate wear and reduce implant lifespan. In cardiovascular devices, mechanical mismatch can affect blood flow dynamics and vessel behavior.
Engineering this interface requires understanding both mechanical and biological responses. Surface treatments, coatings, and structural features are used to improve bonding and reduce relative movement. The goal is not to eliminate interaction, but to ensure that interaction occurs within controlled and predictable limits.
Miniaturization and Emerging Device Challenges
The rise of implantable sensors, neural interfaces, and minimally invasive devices introduces new challenges in mechanical compliance. These devices must function reliably while conforming to highly sensitive and flexible tissues.
Rigid components can cause localized stress or interfere with natural movement. Flexible electronics and soft materials are increasingly being used to address these issues, enabling devices to adapt to the mechanical environment rather than resist it. However, increased flexibility introduces trade-offs in durability and stability. Engineers must balance compliance with the need for long-term functionality, particularly in devices intended for continuous operation.
The Role of Computational Modeling
Advances in computational modeling are enhancing the ability to predict mechanical interactions between implants and biological systems. Finite element analysis and biomechanical simulations allow engineers to evaluate how implants will behave under realistic conditions.
These tools help identify areas of stress concentration, predict long-term deformation, and optimize design before physical implementation. They also support personalized approaches, where implants are tailored to individual patient anatomy and biomechanics. Modeling does not eliminate uncertainty, but it provides a more informed basis for design decisions.
Operational Relevance
The importance of mechanical compliance is becoming more evident as implant technologies advance. Patients are living longer with implanted devices, increasing the demand for durability and sustained performance. At the same time, new applications, such as wearable-integrated implants and bioelectronic systems, are expanding the range of mechanical environments that devices must operate within.
Regulatory expectations are also evolving, with greater emphasis on long-term outcomes rather than initial functionality. This places additional responsibility on engineers to consider how implants perform over extended periods.
System-Level Perspective
Implantable devices cannot be evaluated in isolation. Their performance is defined by how they interact with the human body as a dynamic system. Mechanical compliance matching represents a shift in biomedical engineering from material compatibility to system compatibility. It requires integrating knowledge of mechanics, biology, and long-term behavior into a cohesive design approach.
As biomedical technologies continue to advance, the ability to engineer devices that move, adapt, and respond in harmony with the body will define the next stage of innovation. Stability will not come from rigidity, but from alignment with the system in which the device operates.
